- Home
- Sponsors
- Forums
- Members ˅
- Resources ˅
- Files
- FAQ ˅
- Jobs
-
Webinars ˅
- Upcoming Food Safety Fridays
- Upcoming Hot Topics from Sponsors
- Recorded Food Safety Fridays
- Recorded Food Safety Essentials
- Recorded Hot Topics from Sponsors
- Food Safety Live 2013
- Food Safety Live 2014
- Food Safety Live 2015
- Food Safety Live 2016
- Food Safety Live 2017
- Food Safety Live 2018
- Food Safety Live 2019
- Food Safety Live 2020
- Food Safety Live 2021
- Training ˅
- Links
- Store ˅
- More
Advertisement
Featured Implementation Packages
-
SQF Implementation Package for Food Packaging Manufacturers - Edition 9
This comprehensive documentation package is available for immediate download, ca... more
-
FSSC 22000 Food Safety Management System for Storage & Transport - Version 5.1
This is our premiere package for Food Storage & Distribution Operations look... more
Compressed Air - How Clean is Yours?
Jul 20 2014 07:02 PM | Trace Analytics
compressed air safety purity contamination ISO 8573-1
Understanding compressed air contaminants
ISO 8573-1 identifies the major contaminants of compressed air that can affect the quality in a manufacturing environment where products or machine operation is critical. The standard specifies the levels of allowable contaminants in compressed air in terms of solid matter, water content, and oil content.
Major contaminants as described in ISO 8573-1:
• Particles
• Water
• Oil Aerosol
• Oil Vapor
• Microbial*
*ISO 8573 does not establish limits for microorganisms so we must refer back to environmental testing protocols or in some cases to clean-room specifications.
As can be seen in the table below, ISO 8573 goes well beyond recognized breathing air specifications.
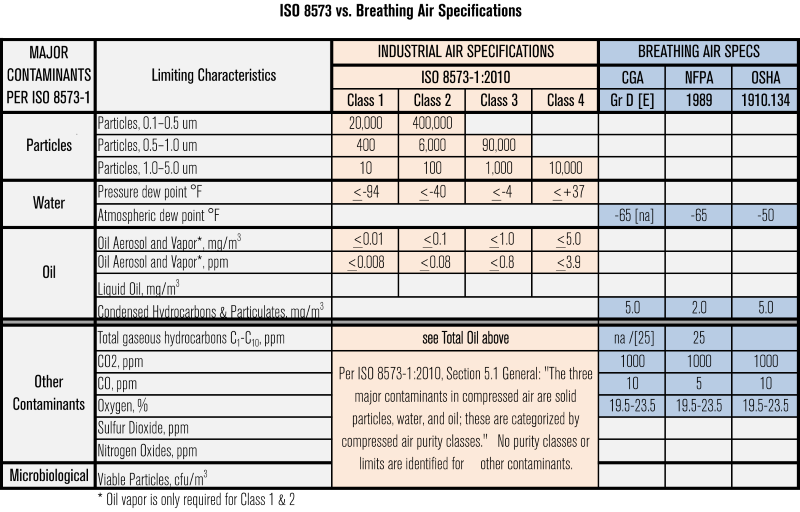
The presence of materials like water, particulate matter, compressor oils and vapors, or microbial contamination in compressed air, used in food processing operations can lead to significant problems. Problems such as:
• Failure of the system
• Product damage
• Increase wastage
• Downtime
• Food quality issues due to out-of specification product (e.g., taste, color, odor)
• Food safety issues due to particulate, oil, or microbial contamination
Avoidance of potential compressed air contaminants
To avoid compressed air contamination, it is important to consider the characteristics of the compressed air required for the operation. It is ideal to consider this during the design phase to ensure system efficiency and stability whilst also ensuring food safety and quality requirements are met.
Great care should also be taken when installing or conducting maintenance on a compressed air system to avoid introducing potential contaminants into the system. New piping should be tested to assure that it has been properly purged of potential contaminants such as particulates, solders, or glues used during installation. Older piping can have an accumulation of water, rust, and oil. When connecting new piping to an older piping distribution system, the jarring of the old piping can cause particulates (such as rust, pipe scale, dirt, metal oxides, etc.) to be loosened and introduced into the new piping. Storage receivers with excess water (vapor, liquid, or a mixture of oil and water) can become a breeding ground for microorganisms.
There exist a number of methods for removing moisture, particulate matter, and oils from compressed air used in food processing activities. These are commonly:
• Separators
• Chemical dryers
• Refrigerators
• Desiccant dryers
• Coalescing Filters
• Sterile Air Filters
• Steam Filters
The choice and level of contaminant mitigation will depend on the range of use of compressed air within the facility. For example, if there is only a slight possibility of incidental contact of compressed air with products and product contact surfaces, then the level of purity requirement will be less than if used for direct steam injection, which is a technique used to warm food and increase the speed of production.
It is wise to seek the advice of commercial filtration and separation specialists in conducting this risk assessment and establishing effective mitigation strategies.
Verifying the purity of compressed air
Once an efficient and effective compressed air system is in operation air quality should be periodically tested at a frequency based on risk assessment. It is best practice to conduct annual or bi-annual testing with additional testing required whenever maintenance work or any activity that may affect the air quality is performed on the compressed air system. Again, if the intended range of uses for compressed air changes then air quality testing may be advisable.
A periodic compressed air test program can provide critical information about your air quality and help prevent contamination of the food supply. Due to the critical nature of compressed air used in the food manufacturing process, qualified personnel should be employed to properly maintain, service, and test the compressed air system.
Trace Analytics offers baseline testing when you are unsure what Purity Classes your air system can meet, they can also provide analysis for any given Purity Classes you select, or you can provide your custom specifications. Trace’s AirCheck Kit™ can be used to sample for particles, water, and oil (aerosol and vapor); Model KPSII is available for microbial sampling. Trace Analytics is an A2LA accredited laboratory in compliance with ISO/IEC 17025. Find out more about compressed air safety in the AirCheck✓™ Academy.










0 Comments