- Home
- Sponsors
- Forums
- Members ˅
- Resources ˅
- Files
- FAQ ˅
- Jobs
-
Webinars ˅
- Upcoming Food Safety Fridays
- Upcoming Hot Topics from Sponsors
- Recorded Food Safety Fridays
- Recorded Food Safety Essentials
- Recorded Hot Topics from Sponsors
- Food Safety Live 2013
- Food Safety Live 2014
- Food Safety Live 2015
- Food Safety Live 2016
- Food Safety Live 2017
- Food Safety Live 2018
- Food Safety Live 2019
- Food Safety Live 2020
- Food Safety Live 2021
- Training ˅
- Links
- Store ˅
- More
Advertisement
Featured Implementation Packages
-
BRCGS Food Safety and Quality Management System for Food Manufacturers - Issue 9
This comprehensive yet simple Food Safety Management System Package contains EVE... more
-
FSSC 22000 Food Safety Management System for Food Manufacturers - Version 6 2025
This is an updated 2025 version of our premiere package for Food Manufacturers l... more
SQF from Scratch: 2.1.4 Complaint Management
Mar 08 2020 02:39 PM | Simon
SQF 2.1.4 Complaint Management
2.1.4 Complaint Management
Complaints happen. No matter how close to perfect a system may be, if the quantities are large enough, even the smallest of mistakes will ultimately reach customers. Thankfully, some of them will take the time to let us know.
While it’s easy to see complaints as just a customer service issue, they also reveal the best opportunities to improve our quality system, both by showing where the problems are happening, and how much the customer cares about the defect. Our best systems will look for metrics while we still have control of the product, but when someone takes the time to pick up the phone or write an email, we need to make sure those individuals aren’t revealing larger problems that have escaped our attention.

The code:
2.1.4 Complaint Management
2.1.4.1 The methods and responsibility for handling and investigating the cause and resolution of complaints from customers and authorities, arising from products manufactured or handled on site, shall be documented and implemented.
2.1.4.2 Trends of customer complaint data shall be investigated and analyzed by personnel knowledgeable about the incidents.
2.1.4.3 Corrective action shall be implemented based on the seriousness of the incident and as outlined in 2.5.3.
2.1.4.4 Records of customer complaints and their investigations shall be implemented.
What’s the point? How is this making our product safer?
Food manufacturing is different than making food at home. If you’re reading this article, you probably already know this. However, it’s not because it’s a business, is subject to regulations, or the products aren’t meals that are eaten immediately.
It’s different because food manufacturers make so much more food than any individual is ever going to make in their lifetime. That means a mistake that might be rare in the home, such as breaking a glass, can become a common occurrence when handling thousands of glasses every day instead of one or two, even if we’re extra careful.
It’s similar to thinking about the lottery. Buying the winning ticket is an uncommon thing, and no one should expect it to happen to them. However, one of the millions of tickets sold winning the lottery is a common occurrence, and should be expected.
Responding to complaint feedback and recording it appropriately allows companies to identify trends that can be traced back to a specific issue.
In the worst-case scenario, practitioners play the role of epidemiologist, identifying a potential outbreak related to the product based on reported illness. Much more commonly, complaint analysis identifies a gap in the system or malfunctioning equipment. Maybe a piece of rubber gasket material that’s been slowing falling apart or a transport issue in the cold supply chain.
Regardless of size or product, every company enjoys the benefit of 100% inspection when the products are finally opened and eaten by the consumer. Complaint management is how we can use that inspection step to verify that our intermittent sampling and inspection activities are doing their job.
What am I being asked to do?
Policy document
To start, there needs to be a policy document in place that identifies the who and how the company responds to complaints and how to determine whether more action is needed. Generally, this will identify the SQF practitioner (or designee) as the one responsible for determining the seriousness of the complaint, and thus the seriousness of the response. This policy/procedure will also define what documentation is used and where it will be kept.
Policy Example:
I. POLICY
- All complaints from customers or authorities that suggest or reflect a failure of product safety shall be entered into the complaint management system.
- Any employee may log a complaint into the system, but only the SQF practitioner or designee can review and close a complaint investigation.
- The SQF Practitioner or designee will use the complaint management system to analyze complaint data for trends, investigate potential root causes, and present data to management.
- Complaints shall be considered valid and initiate investigations/corrective actions if:
- There is enough information to determine where/how the defect may have occurred (e.g. what product, specific descriptions).
- The customer can provide pictures of the affected products or ship them back.
- A food safety or medical authority is involved.
- The complaint is part of a trend or known issue.
- The available information demonstrates the complaint is not a matter of consumer preference or incorrect handling of the product.
- Complaint investigation and corrective actions shall be carried out in response to observed trends and seriousness of the complaint according to our corrective action procedure.
Depending on the product, consumer suspicion can swing wildly from deeply distrustful to “that wouldn’t get me sick in a million years”. The code says the response should be proportionate to the complaint, and that evaluation criteria needs to be defined in the procedure (like in the above example).
No matter what, make sure every complaint is logged for trending, but complaints alleging illness or injury require a more detailed response proportionate to the validity of the complaint. “My stomach hurt after eating your bacon” will demand a different quality of investigation than “my physician believes that this is salmonella, and your product (pictured here) appeared undercooked when I ate it 3 days prior.”
The complaint record
After creating a policy document defining who and how, we also need to include the what. Complaint documentation is going to be used by anyone at the company who might answer the phone and be subject to the wrath of the upset customer, so the form should be intuitive and flexible. Sometimes the only information available is an angry voicemail!
Complaint record example:

Keeping these forms will meet the requirements to have records of customer complaints and the review/actions taken (combined with corrective action records as needed).
Trending
Most public health agencies don’t consider something an outbreak until there are at least two unrelated cases (there are exceptions). So, when a complaint is received, how do we identify if it’s part of a trend or not? This is where the record created above is NOT useful, because a file cabinet full of complaint records does no good.
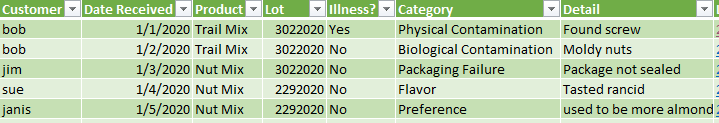
Create a database of some kind that has the critical product information, so that the nature of the complaints can be categorized and presented to management.
This database can be as simple as the date the complaint was received, the product and traceability info, and categorically what the issue was. Complaints of illness or injury should also be called out, as those are going to require more complete investigations.
For a video with detailed instructions on how to create a database like this in excel, click here.
Once a database is made, use graphs and other reporting tools to watch for trends, or simply look at the data and document a narrative of relevant/actionable observations.
Example graph trending by product:
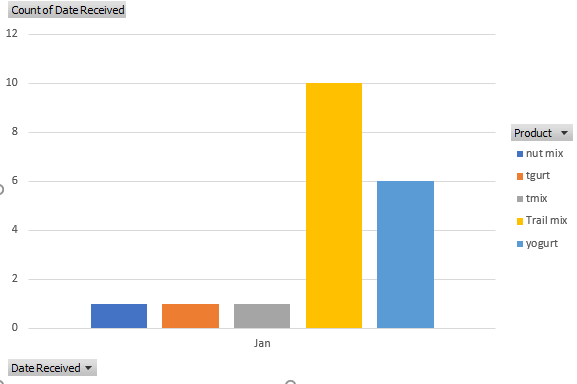
Example trending by narrative:
January 2020
We received 12 complaints in January, most were feedback on the change in almond supplier (more loose skin pieces). However, there was one complaint of a found screw in the product that was determined to be valid. A brief search of our records shows one other screw found in November that looked the same. I have alerted maintenance and sent them the picture and they are examining the trail mix line to determine where the screws are coming from. Will update when we have closed the CAPA.
After reviewing complaints for trends, include the graphs or narrative in a management report and document any observations/corrective actions/investigations that were initiated. Make sure to also close the loop on previous issues and document whether previous investigations/corrections were successful.
For more on how to present complaint trends to management, see SQF From Scratch 2.1.3 Management Review.
How will this be audited?
This and similar programs tend to be audited in this order:
1. Show the policy
2. Show the records
3. Show the management review
4. Show the follow up
The policy review is the strict code audit, is everything there represented in the procedure? Does it have the criteria for “validity” and “seriousness” of the complaint? Who reviews and how?
Next are the records, are they complete, are they readily available, and do they provide useful information. Here it is still helpful to make sure the complaint database doesn’t get polluted with “customer service” type complaints (e.g. billing) as it makes it hard to review for problematic trends.
Auditors will also typically ask for any complaints from authorities (e.g. public health) or complaints of illness specifically, as those will definitely need complete investigations and corrective actions attached. If those investigations are missing, it gives the impression that complaints are just recorded and brushed off as irrelevant or random, which is no good.
Finally, the trending and management communication portion tends to trip up a lot of companies. Many of us cover these items in weekly or monthly meetings verbally, but there needs to be written documentation that these trends were reviewed and it was determined if action was necessary or not. If action was necessary, additional documentation needs to be readily available showing the corrective actions taken. This should fall under the corrective action procedures and be neatly summarized in the management review records.
For a known issue, it can be easy to forget the follow up, or forget to tie it back to the original complaint. Make sure that the CA documentation is duplicated and attached to the complaint, or cross-references it in some other way (e.g. complaint number).
For a smooth audit, identify in advance some recent complaints or complaint trends that were valid, have a complete investigation, and a happy ending. Don’t let an auditor go fishing through the entire complaint history looking for the one they think deserved more attention. Find one that’s relatively harmless, for example a packaging defect, and demonstrate the complete history of the investigation, resolution, and follow up. The goal is to give the auditor confidence that they can “check the box” and think, “yep, I saw all of these code elements in action from start to finish on a complaint that mattered.”
Trivial documentation issues in other complaints will be less of a big deal if the company can demonstrate real and actionable response and follow through on an important one. As with any program audit, if the practitioner can demonstrate that the intent of the requirement is achieved and documented, it is much easier to glide through disagreements in language used or auditor documentation preferences.
2.1.4 is a unique challenge because every facility is going to have a very different “portfolio” of customer complaints. Some are going to have complaints from industry customers in the know, reporting back specification failures and micro results; others will have complaints from concerned parents who report the product is a different color or clumping unusually. They key is to make sure this feedback is seen as valuable, and that we use trending tools to evaluate our system.
Because while the odds of a winner are small, we sell a lot of lottery tickets every day.
Author Biography:

Austin Bouck is a food safety consultant and manufacturing supervisor in Oregon, USA. You can find more food safety resources and discussion on his website, Fur, Farm, and Fork, as well as contact information for consulting services.









0 Comments